Complete Control Plan Leading to the Eradication of Lactoccocus jimmi
Introduction
Pathogen: The deadly pathogen that is causing intracranial suppuration, or brain abscesses, is Lactococcus jimmi. This disease agent was unknown until discovered by our research team. L. jimmi is a Gram positive bacterium (genome size is 4.53 Mb) in the order Lactobacillales (a non-sporeforming order of bacterium) and family Streptococcaceae (family with cocci in chains). The genus Lactococcus does not produce catalsase. L. jimmi does not grow at 40 degrees Celsius, usess arginine as a carbon source, and ferments raffinose with the production of acid.
Reservoir: Aphelocoma californica is commonly known as the western scrub jay. This Avian species can be found in western North America. They can be seen as far north as northern Oregon and as far south as southern Mexico. Their geographical range is from central Texas to the Pacific coast of the United States. The Western scrub jay usually only migrates small distances to lowland areas during winter months (Curry 2002). Their territory size ranges from 6,600 square meters to 65,000 square meters (Sibley 2003). They prefer lower elevations with thick grass, brush, and shrubs. Known predators include raccoons, long-tailed weasels, western, spotted skunks, striped skunks, western gray squirrels, fox squirrels, eastern kingsnakes, pinke snaked, western rattlesnakes, black-billed magpies, American crows, bobcats, Cooper’s hawks, sharp-shinned hawks, golden eagles, red-tailed hawks, and prairie falcons (Clayton 2002). Western scrub jays prefer fruit, nuts, and vegetables, but they also eat small land birds, eggs, California slender salamanders, western fence lizards, and arthropods. Therefor, the western scrub jay is an omniovore. The time of year when scrub jays eat mostly animal matter is during the month of April. The western scrub jay has three known ectoparasite species, all of which are lice: Philopteras cassipes, Brueelia deficienc, and Myrsidea species. They maintain a mutualistic relationship with Odocoileus hemionus columbianus or better commonly known as the Columbian black-tailed deer by eating ticks and flies off of the deer (Isenhart and Desante 1985). They can act as an amplifying host for the transmission of West Nile virus to humans by mosquitoes (Ladeau, Marra, and Calder 2008).
Vector: Ixodes pacificus is commonly known as the western black-legged tick, or the deer tick (CAPC 2015). It is closely related to Ixodes scapularis, which is the deer tick or black-legged tick of the eastern and northern Midwestern United States. I. pacificus belongs to the family Ixodidae, commonly known as hard ticks because of the presence of the scutum, or hard plate, on the dorsal side through all life stages (Molyneux 1993). They are also distinguished by the anterior capitulum that is visible from above (Arnason 1988). With approximately 250 species, the genus Ixodes is the largest in the family Ixodidae. This genus is described as small inornate ticks that lack eyes and festoons with long mouthparts (Taylor, Coop, and Wall 2016). These ticks are relatively small. Female adults average 2.6 mm in length while males about half that size (Furman and Loomis 1984). Red-brown bodies with black legs and capitulum distinguish adult females. Males are usually all black, or rarely, brown. Larvae and nymphs are smaller and have a paler color. Larva are about one-quarter as large as adults. This species of tick is of particular importance in the western United States. It is the main vector of disease that affects both humans and animals on the Pacific coast. There are three active life stages for a tick and they are the larval stage, nymphal stage, and adult stage. There have been 108 vertebrate hosts in the classes of Mammalia, Aves, and Reptilia identified for each of the active life stages of I. pacificus (Castrol and Wright 2007). The stages of the life cycle most likely to bite and infect humans are the nymphs and female adults (CDC 2015). I. pacificus is found in the western or Pacific coast of the United States. Its presence is of particular importance in northern California (CDC 2015). Established populations of I. pacificus were found in 90 counties in five different western states (Dennis, Nekomomoto, Victor, Paul, and Piesman 1998). The geographical distribution of I. pacificus coveres the Pacific Coast of North America to the south coast of British Columbia to southern California and as far inland as southern Utah and Nevada (Arthur and Snow 1968). Over the pas 20 years the geographic range has changed moderately or to a relatively small degree. I. pacificus is already established in most of the counties with a suitable habitat. There are only 11 more western counties that have both suitable climates and surveillance records that indicate that this species is not yet established. This leaves little room for the expansion of the geographical distribution of I. pacificus (Hahn, Jarnevich, Monaghan, and Eisen 2016). This species of ticks can be found in forests, brushy areas, north coastal scrub, and open grasslands. Nymphs can most likely be found on mossy tree trunks. This is due to the moss reducing the surface temperature of the trees and increasing the relative humidity (Slowik and Lane 2001). Nymphs are active from mid-March until mid-August, with their peak density occurring in early May. Nymphal densities correlate positively with rainfall and negatively with maximum air temperatures, typically 23°C and above. This species prefers cooler, moister climatic conditions when compared to warmer, drier conditions (L. Eisen, R. Eisen, and Lane 2002). The most important environmental feature that has an impact on survival is summer desiccation (Bertrand and Wilson 1996). Recently hatched or molted ticks may be able to tolerate drought to a better degree (Padgett and Lane 2001). The life cycle of I. pacificus lasts approximately two years. The life cycle can vary based on different environmental conditions. For example, the life cycle of I. pacificus takes a minimum of three years to complete in northwestern California (Padgett and Lane 2001). There are four stages of the tick life cycle. They are egg, six-legged larva, eight-legged nymph, and adult. The two-year life cycle begins with the eggs typically hatching from late spring to early summer. The ticks are in the larval stage from early summer until the following spring when they become nymphs. These nymphs then become adults in the fall and will lay eggs the following spring. Mating usually takes place on the host, but it can occur off the host as well. After mating the female will detach from the host lay her eggs and then die (NMDH). The number of eggs laid by females ranges from 790 to 1300. The entire egg laying process takes approximately 33 to 40 days (Arthur and Snow 1968). Developmental rates in all three stages are accelerated by warmer soil temperature (Padgett and Lane 2001). Ticks feed by a predatory technique known as questing, which is waiting on the tips of grass or bushes for prey to come along and then latching on. The black-legged tick blood meal usually spans the width of several days (CDC 2011). Female adults typically feed for 10 to 11 days, larvae feed for 4 to 9 days, and nymphs feed for 7 to 11 days (Arthur and Snow 2009). After several days the tick falls off and continues into the next stage of the life cycle. Larva typically feed on small mammals and birds, nymphs feed on small and large mammals, and adults feed on large mammals. Since nymphs are most likely to bite humans, the risk of infection from tick related disease is greatest from late spring to late summer (CDC 2011). I. pacificus is capable of transmitting disease by transovarial transmission and transtadial transmission (Lane and Burgdorfer 1987). This species of ticks has a vector competence of approximately 50% (Lane, Piesman, and Burgdorfer 1991). I. pacificus is known to transmit Lyme disease, Relapsing fever borreliosis, Anaplasmosis and granulocytic ehrlichiosis (HGE). The bacterium responsible for Lyme disease is Borrelia burgdorferi. I. pacificus is responsible for spreading Lyme disease on the Pacific coast of the United States, while Ixodes scapularis is responsible for the spread in the northeast, mid-atlantic, and north-central United States (Braun 2012). A favorite host of I. pacificus is the western fence lizard. This host is reservoir-incompetent for B. borgdorferi which may explain the relatively low prevalence of infection of I. pacificus with B. borgderferi (Lane and Loye 1989). Relapsing fever borreliosis is caused by Borrelia miyamotoi. Anaplasmosis is caused by Anaplasma phagocytophilum. Human granulocytic ehrlichiosis is caused by Erlichia ewingii (CVBD).
Human Host: So far 11 individuals have contracted and died from this emerging disease. Their ages range from 21 years of age to 95 years of age. The age range of people most susceptible to infection is 21 to 70 years of age. All individuals live on the West coast of the United States. The disease originated with patient zero: Kathryn Klein. She most likely contracted the disease while assisting with field research at an archeological site near Xiangtan Mountain Grottoes in Fenfeng, Handan, Heibei China.
Control Design
The design of our control plan begins at the end of the transmission cycle and works backwards. We will first break the link between human host and vector. Then break the transmission link between vector and reservoir. The final step will be to break the link between reservoir and pathogen. With all links being destroyed, the pathogen will eventually cease to proliferate and die off.
Pathogen: The deadly pathogen that is causing intracranial suppuration, or brain abscesses, is Lactococcus jimmi. This disease agent was unknown until discovered by our research team. L. jimmi is a Gram positive bacterium (genome size is 4.53 Mb) in the order Lactobacillales (a non-sporeforming order of bacterium) and family Streptococcaceae (family with cocci in chains). The genus Lactococcus does not produce catalsase. L. jimmi does not grow at 40 degrees Celsius, usess arginine as a carbon source, and ferments raffinose with the production of acid.
Reservoir: Aphelocoma californica is commonly known as the western scrub jay. This Avian species can be found in western North America. They can be seen as far north as northern Oregon and as far south as southern Mexico. Their geographical range is from central Texas to the Pacific coast of the United States. The Western scrub jay usually only migrates small distances to lowland areas during winter months (Curry 2002). Their territory size ranges from 6,600 square meters to 65,000 square meters (Sibley 2003). They prefer lower elevations with thick grass, brush, and shrubs. Known predators include raccoons, long-tailed weasels, western, spotted skunks, striped skunks, western gray squirrels, fox squirrels, eastern kingsnakes, pinke snaked, western rattlesnakes, black-billed magpies, American crows, bobcats, Cooper’s hawks, sharp-shinned hawks, golden eagles, red-tailed hawks, and prairie falcons (Clayton 2002). Western scrub jays prefer fruit, nuts, and vegetables, but they also eat small land birds, eggs, California slender salamanders, western fence lizards, and arthropods. Therefor, the western scrub jay is an omniovore. The time of year when scrub jays eat mostly animal matter is during the month of April. The western scrub jay has three known ectoparasite species, all of which are lice: Philopteras cassipes, Brueelia deficienc, and Myrsidea species. They maintain a mutualistic relationship with Odocoileus hemionus columbianus or better commonly known as the Columbian black-tailed deer by eating ticks and flies off of the deer (Isenhart and Desante 1985). They can act as an amplifying host for the transmission of West Nile virus to humans by mosquitoes (Ladeau, Marra, and Calder 2008).
Vector: Ixodes pacificus is commonly known as the western black-legged tick, or the deer tick (CAPC 2015). It is closely related to Ixodes scapularis, which is the deer tick or black-legged tick of the eastern and northern Midwestern United States. I. pacificus belongs to the family Ixodidae, commonly known as hard ticks because of the presence of the scutum, or hard plate, on the dorsal side through all life stages (Molyneux 1993). They are also distinguished by the anterior capitulum that is visible from above (Arnason 1988). With approximately 250 species, the genus Ixodes is the largest in the family Ixodidae. This genus is described as small inornate ticks that lack eyes and festoons with long mouthparts (Taylor, Coop, and Wall 2016). These ticks are relatively small. Female adults average 2.6 mm in length while males about half that size (Furman and Loomis 1984). Red-brown bodies with black legs and capitulum distinguish adult females. Males are usually all black, or rarely, brown. Larvae and nymphs are smaller and have a paler color. Larva are about one-quarter as large as adults. This species of tick is of particular importance in the western United States. It is the main vector of disease that affects both humans and animals on the Pacific coast. There are three active life stages for a tick and they are the larval stage, nymphal stage, and adult stage. There have been 108 vertebrate hosts in the classes of Mammalia, Aves, and Reptilia identified for each of the active life stages of I. pacificus (Castrol and Wright 2007). The stages of the life cycle most likely to bite and infect humans are the nymphs and female adults (CDC 2015). I. pacificus is found in the western or Pacific coast of the United States. Its presence is of particular importance in northern California (CDC 2015). Established populations of I. pacificus were found in 90 counties in five different western states (Dennis, Nekomomoto, Victor, Paul, and Piesman 1998). The geographical distribution of I. pacificus coveres the Pacific Coast of North America to the south coast of British Columbia to southern California and as far inland as southern Utah and Nevada (Arthur and Snow 1968). Over the pas 20 years the geographic range has changed moderately or to a relatively small degree. I. pacificus is already established in most of the counties with a suitable habitat. There are only 11 more western counties that have both suitable climates and surveillance records that indicate that this species is not yet established. This leaves little room for the expansion of the geographical distribution of I. pacificus (Hahn, Jarnevich, Monaghan, and Eisen 2016). This species of ticks can be found in forests, brushy areas, north coastal scrub, and open grasslands. Nymphs can most likely be found on mossy tree trunks. This is due to the moss reducing the surface temperature of the trees and increasing the relative humidity (Slowik and Lane 2001). Nymphs are active from mid-March until mid-August, with their peak density occurring in early May. Nymphal densities correlate positively with rainfall and negatively with maximum air temperatures, typically 23°C and above. This species prefers cooler, moister climatic conditions when compared to warmer, drier conditions (L. Eisen, R. Eisen, and Lane 2002). The most important environmental feature that has an impact on survival is summer desiccation (Bertrand and Wilson 1996). Recently hatched or molted ticks may be able to tolerate drought to a better degree (Padgett and Lane 2001). The life cycle of I. pacificus lasts approximately two years. The life cycle can vary based on different environmental conditions. For example, the life cycle of I. pacificus takes a minimum of three years to complete in northwestern California (Padgett and Lane 2001). There are four stages of the tick life cycle. They are egg, six-legged larva, eight-legged nymph, and adult. The two-year life cycle begins with the eggs typically hatching from late spring to early summer. The ticks are in the larval stage from early summer until the following spring when they become nymphs. These nymphs then become adults in the fall and will lay eggs the following spring. Mating usually takes place on the host, but it can occur off the host as well. After mating the female will detach from the host lay her eggs and then die (NMDH). The number of eggs laid by females ranges from 790 to 1300. The entire egg laying process takes approximately 33 to 40 days (Arthur and Snow 1968). Developmental rates in all three stages are accelerated by warmer soil temperature (Padgett and Lane 2001). Ticks feed by a predatory technique known as questing, which is waiting on the tips of grass or bushes for prey to come along and then latching on. The black-legged tick blood meal usually spans the width of several days (CDC 2011). Female adults typically feed for 10 to 11 days, larvae feed for 4 to 9 days, and nymphs feed for 7 to 11 days (Arthur and Snow 2009). After several days the tick falls off and continues into the next stage of the life cycle. Larva typically feed on small mammals and birds, nymphs feed on small and large mammals, and adults feed on large mammals. Since nymphs are most likely to bite humans, the risk of infection from tick related disease is greatest from late spring to late summer (CDC 2011). I. pacificus is capable of transmitting disease by transovarial transmission and transtadial transmission (Lane and Burgdorfer 1987). This species of ticks has a vector competence of approximately 50% (Lane, Piesman, and Burgdorfer 1991). I. pacificus is known to transmit Lyme disease, Relapsing fever borreliosis, Anaplasmosis and granulocytic ehrlichiosis (HGE). The bacterium responsible for Lyme disease is Borrelia burgdorferi. I. pacificus is responsible for spreading Lyme disease on the Pacific coast of the United States, while Ixodes scapularis is responsible for the spread in the northeast, mid-atlantic, and north-central United States (Braun 2012). A favorite host of I. pacificus is the western fence lizard. This host is reservoir-incompetent for B. borgdorferi which may explain the relatively low prevalence of infection of I. pacificus with B. borgderferi (Lane and Loye 1989). Relapsing fever borreliosis is caused by Borrelia miyamotoi. Anaplasmosis is caused by Anaplasma phagocytophilum. Human granulocytic ehrlichiosis is caused by Erlichia ewingii (CVBD).
Human Host: So far 11 individuals have contracted and died from this emerging disease. Their ages range from 21 years of age to 95 years of age. The age range of people most susceptible to infection is 21 to 70 years of age. All individuals live on the West coast of the United States. The disease originated with patient zero: Kathryn Klein. She most likely contracted the disease while assisting with field research at an archeological site near Xiangtan Mountain Grottoes in Fenfeng, Handan, Heibei China.
Control Design
The design of our control plan begins at the end of the transmission cycle and works backwards. We will first break the link between human host and vector. Then break the transmission link between vector and reservoir. The final step will be to break the link between reservoir and pathogen. With all links being destroyed, the pathogen will eventually cease to proliferate and die off.
Step 1: Breaking the link between the human host: Vaccination
The only way to initially combat the disease efficiently and effectively would be to create a vaccine that protects against the bacterial agent, L. jimmi, and distribute the vaccine first to those at highest risk of contracting the disease. The main goal in the production and distribution of the vaccine is affordability. Keeping costs down will give more people access to the vaccine and create a broader range of coverage. A broad range of coverage is of utmost importance when dealing with this emerging deadly disease because of the wide distribution of both the vector and reservoir.
Step 2: Breaking the link between vector: Sterile Male Technique
The vector, I. pacificus, will be combated using the sterile male technique. This involves using gamma irradiation on the male tick in order to make him sterile and then releasing them into the wild in order to mate with females. Once the female and the sterile male mates the male usually dies and the female drops off in order to lay her eggs. Since the male was sterile, the female will be unable to lay eggs (Bakirci, Bilgic, Karaer, Duzgun, and Emre 2013). This will lead to the reduction of population size of I. pacificus.
Step 3: Breaking the link between reservoir: Medicated Bird Feeders
In order to eliminate L. jimmi from the reservoir species, A. californica, medicated bird feeders will be placed in the wild. These bird feeders will contain medicated feed that will kill the bacterial pathogen living in the birds that eat out of the feeder (Wallner-Pendleton, Schneider, and Sumner 1993). This will lead to the elimination of L. jimmi in the reservoir species.
The only way to initially combat the disease efficiently and effectively would be to create a vaccine that protects against the bacterial agent, L. jimmi, and distribute the vaccine first to those at highest risk of contracting the disease. The main goal in the production and distribution of the vaccine is affordability. Keeping costs down will give more people access to the vaccine and create a broader range of coverage. A broad range of coverage is of utmost importance when dealing with this emerging deadly disease because of the wide distribution of both the vector and reservoir.
- Creation of Vaccine: The bacterial pathogen that is responsible for this emerging deadly disease is L. jimmi. Vaccines that guard against bacterial pathogens are made in a number of ways, such as identifying the bacterial toxin and making a toxoid, using the polysaccharide of the bacteria, or using two or more proteins from the bacteria (Children’s Hospital of Philadelphia). Any of the previous three routes of making a vaccine would work in the production of the vaccine. In order to proceed efficiently we have decided to utilize the polysaccharide coating in order to create the vaccine. Polysaccharides and proteins add to the complex arrangement of the peptidoglycan layer of gram-positive bacteria (Charpot-Chartier and Kulakauskas 2014). There are three groups of polysaccharides: exopolysaccharides, capsular polysaccharides, and cell wall polysaccharides. Capsular polysaccharides form a shield around the bacterium. Targeting these polysaccharides specifically in the creation of our vaccine will allow us to disrupt the bacterium’s defenses and allow phagocytosis to occur (Bentley, Aanensen, Mavroidi, Saunders, Rabbinowitsch, Collins, Donhoe, Harris, Murphy, Quail, Samuel, Skovsted, Kaltoft, Barrell, Reeves, Parkhill, and Spraft 2006).
- Distribution of Vaccine: Distribution of the vaccine will be based off of three things: age distribution, geographic location of residence, and lifestyle. Individuals of highest concern are those that fit all three of the previously listed criteria: 21 to 70 years of age, live in Thurston, San Louis Obispo, Humboldt, San Francisco, Los Angeles, or Santa Barbara County, and partake in outdoor lifestyles.
- Age Distribution: The age distribution for vaccination is mainly 21 years of age to 70 years of age with mild precautions for those above 70 years of age. In other words children are not our main focus in the distribution of the vaccine. This information is based off the age distribution of patients that have been reported as having contracted the disease.
- Geographic Location of Residence: The vaccine will be distributed to those who reside along the Pacific coast in the Western United States. The states included in this description are Washington, Oregon, and California. The major counties of concern are San Luis Obispo County, Humboldt County, San Francisco County, Los Angeles County, and Santa Barbara County in California and Thurston County in Washington. These counties were identified as the western scrub jay’s hotspots.
- Lifestyle: The vaccine will be distributed to individuals who have an occupation or hobby that places them in areas where the vector, I. pacificus can be found. This includes forested or brushy areas, north coastal scrub, and open grasslands (Slowik and Lane 2001). An example of an occupation that increases the likelihood of contracting the disease is farming. Hobbies include jogging, bird watching, and hiking.
Step 2: Breaking the link between vector: Sterile Male Technique
The vector, I. pacificus, will be combated using the sterile male technique. This involves using gamma irradiation on the male tick in order to make him sterile and then releasing them into the wild in order to mate with females. Once the female and the sterile male mates the male usually dies and the female drops off in order to lay her eggs. Since the male was sterile, the female will be unable to lay eggs (Bakirci, Bilgic, Karaer, Duzgun, and Emre 2013). This will lead to the reduction of population size of I. pacificus.
- Catch: I. pacificus ticks will be caught using the box trap technique used for collecting ticks (Gray 1985). In order to be the most effective, we will use carbon dioxide as the attractant (Carr, Roe, Arellano, Sonenshine, Schal, and Apperson 2013). Female I. pacificus and other species of ticks will be terminated.
- Sterilize: The male I. pacificus ticks will be sterilized using gamma irradiation at the dose of 15 Gray (Gy). This dosage will render the specimens sterile but still allow them to be competitive with wild males (Bakirci, Bilgic, Karaer, Duzgun, and Emre 2013).
- Release: The sterilized males will be released back into the wild. Release points will be widely dispersed in order to effectively eliminate the I. pacificus population.
Step 3: Breaking the link between reservoir: Medicated Bird Feeders
In order to eliminate L. jimmi from the reservoir species, A. californica, medicated bird feeders will be placed in the wild. These bird feeders will contain medicated feed that will kill the bacterial pathogen living in the birds that eat out of the feeder (Wallner-Pendleton, Schneider, and Sumner 1993). This will lead to the elimination of L. jimmi in the reservoir species.
- Creation of Medicated Bird Feed: A medication must be created in order to kill the L. jimmi pathogen living in A. californica. This medication will be applied / mixed in to birdseed.
- Distribution of Medicated Bird Feed: The medicated bird feed will be distributed at pet and garden centers in the six hotspot counties for locals to pickup and place in their home feeders. Our team of researchers will also place feeders in public locations such as community parks.
Implementation of Control
The control plan will be implemented in a series of phases. Although these phases are numbered, they are not sequential in time. The phases will overlap. The phase number corresponds to importance and there priority for initiation.
Phase 1: Creation and Distribution of Vaccine
Phase 2: Catch, Sterilize, Release of Male Ixodes pacificus
Phase 3: Medicated Bird Feeders
Potential Problems
It has been noted that only 33% of people accept the vaccine due to safety concerns. Further studies are being conducted on safety and these findings will be issued as soon as available. For now, our research team is issuing a public health announcement stating that the vaccine has shown no signs of harm in patients who have already received the vaccine. The vaccine has shown to be 94% effective. Patients who receive the vaccine report side effects. We will continue to research the development of a vaccine that is more effective and produces fewer side effects. In response to the Vaccination Liberation group’s press conference and letters, we will hold a press conference of our own and express the effectiveness of the vaccination along with the number of new cases (6,531) and cases resulting in death (653). The petition for the recall of the vaccination will be ignored for now. There have been no further issues reported at this time. Further issues will be addressed as they occur.
Expected Outcomes
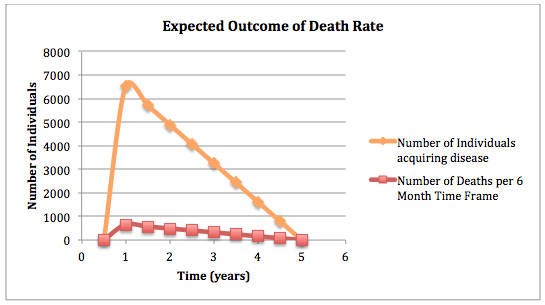
It is expected that we will see the eradication of L. jimmi within the next 5 years if all the above steps are taken and followed correctly and efficiently. The effective eradication will require a community wide effort. Individuals will need to acquire immunizations and place medicated bird feeders at their residences.
The control plan will be implemented in a series of phases. Although these phases are numbered, they are not sequential in time. The phases will overlap. The phase number corresponds to importance and there priority for initiation.
Phase 1: Creation and Distribution of Vaccine
- Materials: There are approximately 5,255,112 individuals living in the hotspot counties of Thurston, San Louis Obispo, Humboldt, San Francisco, Los Angeles, and Santa Barbara (US Census Bureau 2013). The percent of people in these counties that are in the age range from 21 to 70 is approximately 64% (Wikipedia). The percent of people in the United States that participate in some form of outdoors activity is 49.4% (The Outdoor Foundation 2013). The total number of single dose vaccine vials needed is approximately 1,700,000. This number is based off the total population in the hotspot counties (5,255,112) times the percent of people that fall in the required age group (64%) times the percent of people who participate in some form of outdoor recreation (49.4%). The same number (1,700,000) of 3 mL 1.5 inch needle syringes will be needed.
- Methods: The vaccine will be created using the polysaccharide coating as the basis of the vaccination. The vials will then be filled with the vaccine and distributed. Distribution will take place at local clinics and hospitals in the six major counties: San Luis Obispo County, Humboldt County, San Francisco County, Los Angeles County, and Santa Barbara County in California and Thurston County in Washington. Television commercials, radio announcements, and flyers will be used to spread the word about the impeding threat of the emerging deadly disease and the vaccine as a solution. The vaccine will be distributed in three sub-phases. Sub-phase one will distribute vaccines to only the individuals who meet the criteria: 21 to 70 years of age, live in Thurston, San Louis Obispo, Humboldt, San Francisco, Los Angeles, or Santa Barbara County, and partake in outdoor lifestyles. Sub-phase one will last approximately one week then phase two will begin. Sub-phase two will distribute vaccinations people of all ages living in the six major counties and partaking in outdoor lifestyles. Sub-phase two will last approximately one week then phase three will begin. Sub-phase three will allow anyone, regardless of age, geographic location, and lifestyle, to obtain the vaccination.
Phase 2: Catch, Sterilize, Release of Male Ixodes pacificus
- Materials: In order to make the box traps we will need carbon dioxide attractant, a box large enough to contain attractant, an angled platform, sticky tape, weather protective covering, and ground stakes. We will need enough supplies to place a trap in each of the counties of primary concern: San Luis Obispo County, Humboldt County, San Francisco County, Los Angeles County, and Santa Barbara County in California and Thurston County in Washington. Once the ticks are caught we will need transport containers and a gamma irradiation machine to sterilize the male I. pacificus ticks.
- Methods: All six traps will be constructed by placing the sticky tape on the top side of the platform and surrounding the box with the platform, and then covering it to keep it safe from the weather. One trap will be placed in each of the six hotspot counties. The traps will be placed in brushy areas and baited with carbon dioxide attractant. The trap will be staked into the ground in order to prevent it from blowing away or moved by animals. Each week the traps will be checked, specimens collected, and re-baited. The female I. pacificus ticks and other species of ticks will be terminated. Only the male I. pacificus ticks will be placed in transport containers and returned to the lab in order to be sterilized. A gamma irradiation machine will be used to deliver 15 Gray to each male tick. These sterilized males will then be returned to the six hotspot counties but will be dispersed to at least five different locations within each county. They will be dispersed in brushy areas no less than one mile from the traps in order to prevent the sterilized males from being recaptured.
Phase 3: Medicated Bird Feeders
- Materials: In order to eliminate L. jimmi from the A. californica population we will need medication that is capable of killing the bacterial pathogen without harming the reservoir. Once this medication is created, we will need bird seed to apply the medication to. The bird seed will be need to be packaged. Our research team will need to aquire approximately 60 bird feeders in order to place the medicated bird feed throughout hotspot counties.
- Methods: The medicated bird feed will be created and distributed to pet and garden centers in the hotspot counties. Locals will be informed of the bird feed through television commercials, radio announcements, and flyers. Locals will have the opportunity to obtain and fill their home feeders with the medicated bird feed in order to guard against the disease. Our research team will also be filling feeders and distributing them throughout the hot spot counties. We will start by placing the feeders in parks and on running/hiking trails. These feeders will be monitored and refilled as necessary.
Potential Problems
It has been noted that only 33% of people accept the vaccine due to safety concerns. Further studies are being conducted on safety and these findings will be issued as soon as available. For now, our research team is issuing a public health announcement stating that the vaccine has shown no signs of harm in patients who have already received the vaccine. The vaccine has shown to be 94% effective. Patients who receive the vaccine report side effects. We will continue to research the development of a vaccine that is more effective and produces fewer side effects. In response to the Vaccination Liberation group’s press conference and letters, we will hold a press conference of our own and express the effectiveness of the vaccination along with the number of new cases (6,531) and cases resulting in death (653). The petition for the recall of the vaccination will be ignored for now. There have been no further issues reported at this time. Further issues will be addressed as they occur.
Expected Outcomes
It is expected that we will see the eradication of L. jimmi within the next 5 years if all the above steps are taken and followed correctly and efficiently. The effective eradication will require a community wide effort. Individuals will need to acquire immunizations and place medicated bird feeders at their residences.
For now, the number of deaths due to this horrendous disease will continue to rise. At the 1-year mark we will begin to see a decrease in deaths due to immunization effectiveness, vector elimination, and pathogen eradication.
The above protocol should be continued even after it is believed that L. jimmi has been eradicated in order to prevent another relapse of a potentially more resistant L. jimmi species. If the protocol is discontinued then L. jimmi may acquire a resistance that will be even harder to combat in the future. If the bacterium was to reemerge into human populations then effectiveness of the above protocol could be doubled (distribute more vaccines, sterilize more male I. pacificus, and distribute more medicated bird feed). Further steps may need to be taken as well. Such steps may include closing community parks and hiking trails. These issues will be discussed and addressed as the need arises.
References
Arnason C. S. 1992. Biology of the Western Black-legged Tick, Ixodes pacificus, (Cooley and Kohls, 1943): A Potential Vector of Lyme Disease in South Coastal British Columbia. <summit.sfu.ca/system/files/iritems1/3850/b1426397x.pdf>.
Arthur, D. R. and K. R. Snow. 1968. Ixodes pacificus Cooley and Kohls, 1943: Its life-history and occurrence. Parasitol. 58:893-906. Web.
Bakirci, S., H. B. Bilgic, Z. Karear, A. Duzgun, and Z. Emre. 2013. Studies on the Application of the Sterile-Male Technique on the Tick Hyalomma Excavatum. Veteriner Fakultesi dergisi. 60(2):93-98. Web.
Braun D. M. 2012. Disease-Spreading Ticks on the Move as Climate Changes. National Geographic. < http://voices.nationalgeographic.com/2012/09/07/disease-spreading-ticks-on-the-move-as-climate-changes/>.
Bentley SD, Aanensen DM, Mavroidi A, Saunders D, Rabbinowitsch E, Collins M, Donohoe K, Harris D, Murphy L, Quail MA, Samuel G, Skovsted IC, Kaltoft MS, Barrell B, Reeves PR, Parkhill J, Spratt BG. 2006. Genetic Analysis of the Capsular Biosynthetic Locus from all 90 Pneumococcal Serotypes. PLoS Genet. 10.1371/journal.pgen.0020031. Web.
Bertrand, M. R. and M. I. Wilson. 1996. Microclimate-dependent Survival of Unfed Adult Ixodes scapularis (Acari: Ixodidae) in Nature: Life Cycle and Study Design Implications. J Med Entomol. 33:619-627.
(CAPC) Companion Animal Parasite Council. 2015. Vector-Borne Diseases – Ixodes Scapularis and Ixodes Pacificus. <https://www.capcvet.org/capc-recommendations/ixodes-scapularis-and-ixodes-pacificus>.
Carr, A. L., R. M. Roe, C. Arellano, D. E. Sonenshine, C. Schal, and C. S. Apperson. 2013. Responses of Amblyomma Americanum and Dermacentor Variabilis to Odorants That Attract Haematophagous Insects. Med and Vet Entomol. 27.1 (2012): 86-95. Web.
Casrtol, M. B., and S. A. Wright. 2007. Vertebrate hosts of Ixodes pacificus (Acari: Ixodidae) in California. J Vector Ecol. 32(1):140-149. Web.
(CDC) Center for Disease Control and Prevention. 2015. Geographic Distribution of Ticks that Bite Humans. <http://www.cdc.gov/ticks/geographic_distribution.html>.
(CDC) Center for Disease Control and Prevention. 2011. Lifecycle of Blacklegged Ticks. <https://www.cdc.gov/lyme/transmission/blacklegged.html>.
Chapot-Chartier, M-P and S. Kulakauskas. 2014. Cell Wall Structure and Function of Lactic Acid Bacteria. Microb Cell Fact. 10.1186/1475-2859-13-S1-S9. Web.
Children’s Hospital of Philadelphia. Making Vaccines: How are Vaccines Made?. http://www.chop.edu/centers-programs/vaccine-education-center/making-vaccines/how-are-vaccines-made.
Clayton, D. 2002. Influence of Bill Shape on Ectoparasite Load in Wester Scrub-jays. The Condor. 104:675-678.
Curry, R. 2002. Western Scrub-jay: Aphelocoma californica. Phiadelphia, Pa.: Birds of North America.
(CVBD) Companion Vector-Borne Diseases. Ehrlichiosis. < http://www.cvbd.org/en/tick-borne-diseases/ehrlichiosis/pathogens/>.
Dennis, D. T., T. S. Nekomoto, J. C. Victor, W. S. Paul, and J. Piesman. 1998. Forum: Reported Distribution of Ixodes scapularis and Ixodes pacificus (Acari: Ixodidae) in the United States. J Med Entomol. 35(5):629-638. Web.
Eisen, L., R. J. Eisen, R. S. Lane. 2002. Seasonal Activity Patterns of Ixodes pacificus Nymphs in Relation to Climatic Conditions. Medical and Veterinary Entomology, 16, 235-244. 10.1046. Web.
Furman, D. P. and E. C. Loomis. 1984. The ticks of California, Bull. Calif Insect Surv. Vol.25, 356 pp.
Gray, J. S. 1985. A Carbon Dioxide Trap for Prolonged Sampling OfIxodes Ricinus L. Populations. Experimental & Applied Acarology 1.1 (1985): 35-44. Web.
Hahn, M. B., C. S. Jarnevich, A. J. Monaghan, and R. J. Eisen. 2016. Modeling the Geographic Distribution of Ixodes scapularis and Ixodes pacificus (Acari: Ixodidae) in the Contiguous United States. J Med Entomol. 1176-1191. Web.
Isenhart, F. and D. Desante. 1985. Observations of Scrub Jays Cleaning Ectoparasites from Black-tailed Deer. The Condor. 87:145-147.
Ladeau, S., P. Marra, K. A, and C. Calder. 2008. West Nile Virus Revisted: Consequences for North American Ecology. Bioscience. 58/10:937-946.
Lane, R. S. and W. Burgdorfer. 1987. Transovarial and transtadial passage of Borrelia burgdorferi in the Western Black-legged Tick, Ixodes pacificus (Acari: Ixodidae). Am J Trop Med Hyg. 37:188-192.
Lane, R. S., and J. E. Loye. 1989. Lyme Disease in California: Interrelationship of Ixodes pacificus (Acari: IIxodidae), the Western Fence Lizard (Sceloporus occidentalis), and Borrelia burgdorferi. J Med Entomol. 26(4):272-8. Web.
Lane, R. S., J. Piesman, and W. Burgdorfer. 1991. Lyme Borreliosis: Relation of its Causative Agent to its Vectors and Hosts in North America and Europe. Ann Rev Entomol. 36:587-609.
Molyneux, D. H. 1993. Vectors. Modern parasitology: a textbook of parasitology. Pp. 53-74. ISBN 978-0-632-02585-5.
(NMDH) New Mexico Department of Health. New Mexico Vector Control Manual. < https://nmhealth.org/>.
The Outdoor Foundation. 2013. Outdoor Participation Report. www.outdoorfoundation.org/pdf/ResearchParticipation2013.pdf.
Padgett, K. A. and R. S. Lane. 2001. Life Cycle of Ixodes pacificus (Acari: Ixodidae): Timing of Developmental Processes Under Field and Laboratory Conditions. J Med Entomol. 38:684-693. Web.
Sibley, D. 2003. The Sibley Field Guide to Birds. New York, United States: Alfred A. Knopf, Inc.
Slowik, T. J. and R. S. Lane. 2001. Nymphs of the Western Black-legged tick (Ixodes pacificus) Collected from Tree Trunks in Woodland-grass Habitat. J Vector Ecol. 26(2): 165-171.
Taylor, M. A., R. L. Coop, and R. L. Wall. 2016. Veterinary Parasitology. Pp. 242-245. ISBN 978-0-470-67162-7.
U. S. Census Bureau. 2013. Population Estimates. http://www.census.gov/popest/about/terms.html.
Wallner-Pendleton, E., N. Schneider, and S. Sumner. 1993. G93-1147 Preventing Bacterial Contamination, Medication and Other Chemical Residues in Poultry Meat and Eggs. Historical Materials from University of Nebraska-Lincoln Extension. Paper 1288.
Arnason C. S. 1992. Biology of the Western Black-legged Tick, Ixodes pacificus, (Cooley and Kohls, 1943): A Potential Vector of Lyme Disease in South Coastal British Columbia. <summit.sfu.ca/system/files/iritems1/3850/b1426397x.pdf>.
Arthur, D. R. and K. R. Snow. 1968. Ixodes pacificus Cooley and Kohls, 1943: Its life-history and occurrence. Parasitol. 58:893-906. Web.
Bakirci, S., H. B. Bilgic, Z. Karear, A. Duzgun, and Z. Emre. 2013. Studies on the Application of the Sterile-Male Technique on the Tick Hyalomma Excavatum. Veteriner Fakultesi dergisi. 60(2):93-98. Web.
Braun D. M. 2012. Disease-Spreading Ticks on the Move as Climate Changes. National Geographic. < http://voices.nationalgeographic.com/2012/09/07/disease-spreading-ticks-on-the-move-as-climate-changes/>.
Bentley SD, Aanensen DM, Mavroidi A, Saunders D, Rabbinowitsch E, Collins M, Donohoe K, Harris D, Murphy L, Quail MA, Samuel G, Skovsted IC, Kaltoft MS, Barrell B, Reeves PR, Parkhill J, Spratt BG. 2006. Genetic Analysis of the Capsular Biosynthetic Locus from all 90 Pneumococcal Serotypes. PLoS Genet. 10.1371/journal.pgen.0020031. Web.
Bertrand, M. R. and M. I. Wilson. 1996. Microclimate-dependent Survival of Unfed Adult Ixodes scapularis (Acari: Ixodidae) in Nature: Life Cycle and Study Design Implications. J Med Entomol. 33:619-627.
(CAPC) Companion Animal Parasite Council. 2015. Vector-Borne Diseases – Ixodes Scapularis and Ixodes Pacificus. <https://www.capcvet.org/capc-recommendations/ixodes-scapularis-and-ixodes-pacificus>.
Carr, A. L., R. M. Roe, C. Arellano, D. E. Sonenshine, C. Schal, and C. S. Apperson. 2013. Responses of Amblyomma Americanum and Dermacentor Variabilis to Odorants That Attract Haematophagous Insects. Med and Vet Entomol. 27.1 (2012): 86-95. Web.
Casrtol, M. B., and S. A. Wright. 2007. Vertebrate hosts of Ixodes pacificus (Acari: Ixodidae) in California. J Vector Ecol. 32(1):140-149. Web.
(CDC) Center for Disease Control and Prevention. 2015. Geographic Distribution of Ticks that Bite Humans. <http://www.cdc.gov/ticks/geographic_distribution.html>.
(CDC) Center for Disease Control and Prevention. 2011. Lifecycle of Blacklegged Ticks. <https://www.cdc.gov/lyme/transmission/blacklegged.html>.
Chapot-Chartier, M-P and S. Kulakauskas. 2014. Cell Wall Structure and Function of Lactic Acid Bacteria. Microb Cell Fact. 10.1186/1475-2859-13-S1-S9. Web.
Children’s Hospital of Philadelphia. Making Vaccines: How are Vaccines Made?. http://www.chop.edu/centers-programs/vaccine-education-center/making-vaccines/how-are-vaccines-made.
Clayton, D. 2002. Influence of Bill Shape on Ectoparasite Load in Wester Scrub-jays. The Condor. 104:675-678.
Curry, R. 2002. Western Scrub-jay: Aphelocoma californica. Phiadelphia, Pa.: Birds of North America.
(CVBD) Companion Vector-Borne Diseases. Ehrlichiosis. < http://www.cvbd.org/en/tick-borne-diseases/ehrlichiosis/pathogens/>.
Dennis, D. T., T. S. Nekomoto, J. C. Victor, W. S. Paul, and J. Piesman. 1998. Forum: Reported Distribution of Ixodes scapularis and Ixodes pacificus (Acari: Ixodidae) in the United States. J Med Entomol. 35(5):629-638. Web.
Eisen, L., R. J. Eisen, R. S. Lane. 2002. Seasonal Activity Patterns of Ixodes pacificus Nymphs in Relation to Climatic Conditions. Medical and Veterinary Entomology, 16, 235-244. 10.1046. Web.
Furman, D. P. and E. C. Loomis. 1984. The ticks of California, Bull. Calif Insect Surv. Vol.25, 356 pp.
Gray, J. S. 1985. A Carbon Dioxide Trap for Prolonged Sampling OfIxodes Ricinus L. Populations. Experimental & Applied Acarology 1.1 (1985): 35-44. Web.
Hahn, M. B., C. S. Jarnevich, A. J. Monaghan, and R. J. Eisen. 2016. Modeling the Geographic Distribution of Ixodes scapularis and Ixodes pacificus (Acari: Ixodidae) in the Contiguous United States. J Med Entomol. 1176-1191. Web.
Isenhart, F. and D. Desante. 1985. Observations of Scrub Jays Cleaning Ectoparasites from Black-tailed Deer. The Condor. 87:145-147.
Ladeau, S., P. Marra, K. A, and C. Calder. 2008. West Nile Virus Revisted: Consequences for North American Ecology. Bioscience. 58/10:937-946.
Lane, R. S. and W. Burgdorfer. 1987. Transovarial and transtadial passage of Borrelia burgdorferi in the Western Black-legged Tick, Ixodes pacificus (Acari: Ixodidae). Am J Trop Med Hyg. 37:188-192.
Lane, R. S., and J. E. Loye. 1989. Lyme Disease in California: Interrelationship of Ixodes pacificus (Acari: IIxodidae), the Western Fence Lizard (Sceloporus occidentalis), and Borrelia burgdorferi. J Med Entomol. 26(4):272-8. Web.
Lane, R. S., J. Piesman, and W. Burgdorfer. 1991. Lyme Borreliosis: Relation of its Causative Agent to its Vectors and Hosts in North America and Europe. Ann Rev Entomol. 36:587-609.
Molyneux, D. H. 1993. Vectors. Modern parasitology: a textbook of parasitology. Pp. 53-74. ISBN 978-0-632-02585-5.
(NMDH) New Mexico Department of Health. New Mexico Vector Control Manual. < https://nmhealth.org/>.
The Outdoor Foundation. 2013. Outdoor Participation Report. www.outdoorfoundation.org/pdf/ResearchParticipation2013.pdf.
Padgett, K. A. and R. S. Lane. 2001. Life Cycle of Ixodes pacificus (Acari: Ixodidae): Timing of Developmental Processes Under Field and Laboratory Conditions. J Med Entomol. 38:684-693. Web.
Sibley, D. 2003. The Sibley Field Guide to Birds. New York, United States: Alfred A. Knopf, Inc.
Slowik, T. J. and R. S. Lane. 2001. Nymphs of the Western Black-legged tick (Ixodes pacificus) Collected from Tree Trunks in Woodland-grass Habitat. J Vector Ecol. 26(2): 165-171.
Taylor, M. A., R. L. Coop, and R. L. Wall. 2016. Veterinary Parasitology. Pp. 242-245. ISBN 978-0-470-67162-7.
U. S. Census Bureau. 2013. Population Estimates. http://www.census.gov/popest/about/terms.html.
Wallner-Pendleton, E., N. Schneider, and S. Sumner. 1993. G93-1147 Preventing Bacterial Contamination, Medication and Other Chemical Residues in Poultry Meat and Eggs. Historical Materials from University of Nebraska-Lincoln Extension. Paper 1288.
Your browser does not support viewing this document. Click here to download the document.